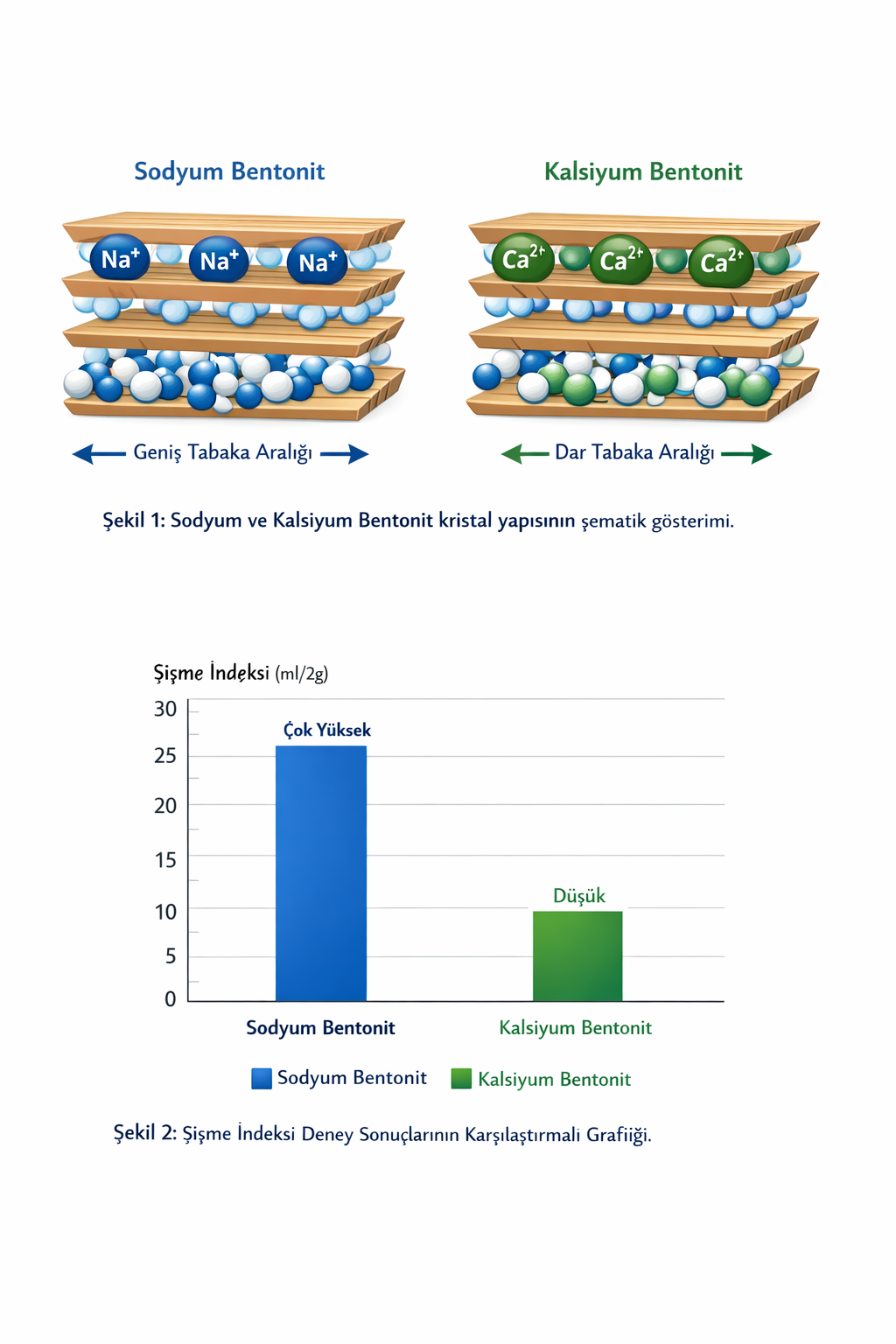
1. Mineralogical and Crystal Chemical Basis of Bentonite
Bentonite is a 2:1 type layered silicate clay mineral formed by hydrothermal alteration of volcanic tuffs, with montmorillonite as its primary mineral. Bentonites are classified according to the nature of exchangeable cations in the interlayer space as sodium (Na⁺) or calcium (Ca²⁺) saturated.
1.1. Crystal Structure and Cation Exchange Capacity
Montmorillonite structure consists of an aluminum octahedral sheet sandwiched between two silicon tetrahedral sheets. Isomorphic substitutions in tetrahedral sheets create net negative surface charge.
Key distinguishing properties of sodium and calcium bentonites:
1.2. Colloidal and Surface Chemistry
Colloidal behavior of bentonites can be explained by DLVO theory. In sodium bentonite, monovalent Na⁺ ions attract more water molecules into the interlayer space, increasing zeta potential (-30mV to -50mV).
2. Industrial Applications and Comparative Analysis
Sodium and calcium bentonites are preferred in different industrial applications due to differences in their physicochemical properties.
2.1. Drilling Industry (Drilling Muds)
2.2. Foundry and Metallurgy Industry
2.3. Construction and Geotechnical Applications
2.4. Food, Cosmetics and Pharmaceutical
3. Comprehensive Laboratory Test Methods
The following standard test methods are applied for quality control of sodium and calcium bentonites.
3.1. Swelling Index Test (ASTM D5890)
Purpose: Water absorption and volumetric expansion capacity.
Procedure: 2.00±0.01 g bentonite + 100 mL deionized water. Stand at 25±2°C for 2 hours.
Evaluation: Sodium ≥25 mL/2g, Calcium 5-15 mL/2g, Activated 20-28 mL/2g.
3.2. Methylene Blue Test (ASTM C837)
Purpose: Clay content and CEC determination.
Calculation: MBT (mL/100g) = (Methylene volume / Sample weight) × 100
Result: Sodium 35-50, Calcium 20-35 mL/100g.
3.3. Viscosity and Rheology (API RP 13B-1)
Sample: 22.5 g bentonite + 350 mL water. 16-24 hours hydration.
Calculations:
• PV = θ₆₀₀ - θ₃₀₀ (cP)
• YP = θ₃₀₀ - PV (lb/100ft²)
• YP/PV ≤3 (API limit)
Required: PV ≥4 cP, 10-sec gel ≥3, 10-min gel ≤32.
3.4. Fluid Loss Test
API Filter Press: 100 psi, 30 min, 25°C.
Limit: API 13A ≤15 mL/30 min. High quality ≤12 mL.
3.5. Foundry Performance Tests
Wet Tensile: Sodium ≥3.5, Calcium ≥2.0 N/cm²
Dry Tensile: Sodium ≥10, Calcium ≥6 N/cm²
Blue Index: Sodium ≥35, Calcium 25-35 mL.
4. Bentonite Selection Decision Tree
Yes → Sodium Bentonite | No → Calcium Bentonite
Yes → Modified Sodium | No → Standard Sodium
• Oil Drilling: API Sodium
• Iron Foundry: Sodium
• Aluminum: Calcium
• Slurry Walls: High Viscosity Sodium
• Animal Feed: Calcium
• Cosmetics: Purified Sodium
5. Conclusion and Academic Evaluation
Selection of sodium and calcium bentonites in industrial applications requires holistic evaluation of crystal chemistry, colloidal behavior, and specific performance requirements. Sodium bentonite is preferred for high-performance applications due to its high swelling index and viscosity development capacity, while calcium bentonite is valued for low-temperature applications due to its economic advantage.
Activation of calcium bentonite with sodium carbonate is an important process, but use of natural sodium bentonite compliant with API/OCMA standards is recommended for critical applications. Application of laboratory tests according to standard procedures is essential for quality control.
Supply and Industrial Cooperation
The technical data, API/OCMA standard analyses, and industrial application examples presented in this academic study were prepared using Miner Mining (Nevşehir) company's bentonite product range and quality control laboratory data.
For high-quality certified bentonite supply and technical support:
www.miner.com.tr
References and Standards
- API Specification 13A, 18th Edition, American Petroleum Institute, 2010.
- API Recommended Practice 13B-1, American Petroleum Institute, 2003.
- ASTM D5890, "Swelling Index of Clay Mineral Component of GCLs".
- ASTM C837, "Methylene Blue Index of Clay".
- OCMA Specification DFCP-4, "Drilling Grade Bentonite", 1983.
- Murray, H.H., "Applied Clay Mineralogy", Elsevier, 2007.
- Bergaya, F., Lagaly, G., "Handbook of Clay Science", Elsevier, 2013.
- Caenn, R., et al., "Composition and Properties of Drilling Fluids", 7th Ed., 2017.
- Odom, I.E., "Smectite Clay Minerals: Properties and Uses", 1984.
- Karakaya, N., Kocabaş, S., "Activation of Bentonites", Journal of Mining, 2006.
© 2026 bentonit.net.tr publication. All rights reserved.